Slag analysis – Schlackologie

Slag samples of a medieval (12th century) copper smelting furnace.
Top left: #21, XPL. Under crossed polarisers the zinc sulphide can be seen as tiny dendrites within the glassy matrix. The other phases are willemite (wlm) and copper (Cu), which is usually asso ciated with the (Zn,Fe,Cu)S phase, an accompanying phase of copper matte at the Huneberg, from which it probably segregates upon solidification. Free iron oxides are seen as dark and much larger dendrites.
Top right: Another section of #21, under plain polarised light. It shows the abundant spinels, the iron oxides and copper inclusions. Most notably it shows the free baryte (brt) inclusion in the slag matrix.
Bottom left: #22, shows more free baryte (brt), together with a lead antimony matte. The long needles are a mixed iron and zinc oxides and are believed to be franklinite (frk?). #104, is rather more affected by corrosion than the other two samples. It shows mainly spinel (sp), free iron oxides and some willemite (wlm). Spinels are in the same order of magnitude than in the other samples.
The colloquial term Schlackologie is used by several German colleagues in the archaeometric sciences when it comes to the analysis and interpretation of mostly metallurgical slags. Literally it would translate to slagology. But why are slags investigated at all and what exactly is slag?
What are slags and what do they consist of?
Slags are by-products of metallurgical or pyro-technological processes and usually remain at the production site as waste product. For example metallurgical smelting slag is usually composed of five separate components:
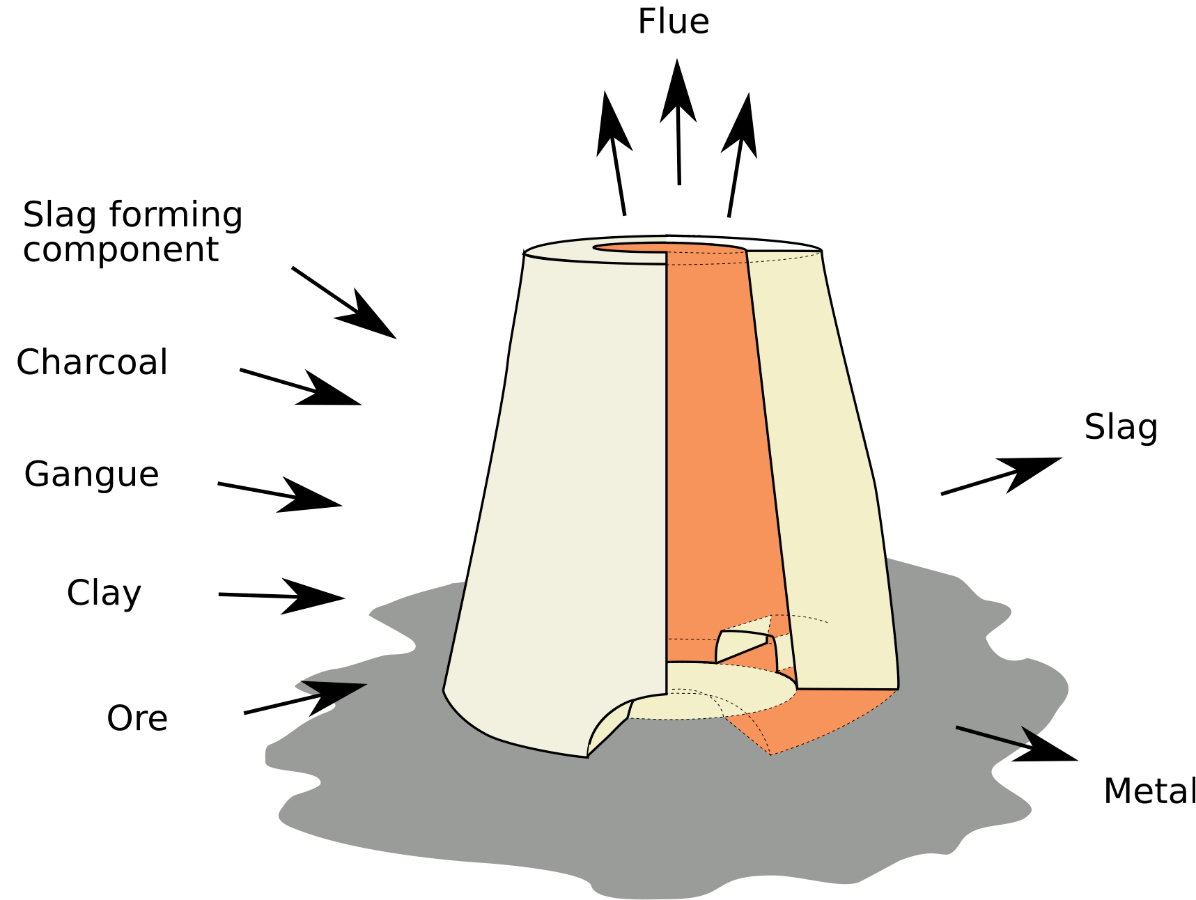
Schema of a smeting furnaces, its charge ingredients and its waste-, by- and end products
- Ore
- Gangue
- Fuel ash
- Furnace construction materials
- Slag forming agent / flux
In theory slag is supposed to collect all the undesired ingredients of the furnace charge, leaving the desired metal behind. It is furthermore desired that slag and metal may be easily separated from the metal. To meet this requirement slag should possess a low melting point and low viscosity. In practice these requirements are not always met and tell us a lot about the processes and capabilities of the ancient metal workers.
There are ores that already contain the necessary chemical compounds tor produce a low melting and low viscous slag, however as to whether fluxing happened intentionally or not may in most cases only be addressed towards the end of the analytical process.
Slags are often found in large amounts at smelting sites and show the extent of metal production. Due to the large amounts they are often taken only as samples during archaeological digs.
Slags may appear from glassy to bubbly from frothy to compact to and from homogeneous to heterogeneous, and their colour ranges from black to brown or grey. In some cases there may also be green or blue slag. A macroscopic distinction between smelting slags, furnace slag, smithing slag, or vitrified furnace building materials (clay, loam, brasque) is not always possible and requires further investigation.

Medieval copper smelting slag. Macroscopically different kinds of slag need not necessarily possess a different chemical composition.
As a rule slags consist of silicates, ie compounds in which silica is necessary for their formation.The majority of the archaeologically recorded slags possess fayaliteFe2SiO4 as the main ingredient which forms at a particularly low temperature in the SiO2 – FeO – Al2O3 system. It makes no difference as to whether this is iron- or sulfidic copper ore smelting.
Why is slag investigated?
There are a variety of processes in which slags are produced; a few are mentioned above. To determine which process was practised on site, slags may be investigated. By their phase constituents and their chemical composition the conditions of their formation can be reconstructed. Slag acts as a process log of chemical and pyro-technological processes. We are able to access and interpret this information. The quantity of slag on a site may also be used to quantify the metal production output.
What information can be expected from slag?
Individual phases within slag form under physical conditions such as pressure, temperature and furnace atmosphere (the so-called partial oxygen pressure or oxygen fugacity). These physical conditions do not change over time. Fayalite forms under the same conditions now than 2000 years ago. The variability of slag is a result of the chemical compounds which participated during slag formation. We can reconstruct these physical conditions by examining the phase composition of slag by microscopical means. The chemical composition, or rather the bulk chemical composition is determined and is used to answer questions with regard to the nature of furnace charge such as: Which ores were used? Was there any intentional flux added to the charge? Where are the clay and other raw material sources?
How is slag investigated?
Slag has to be investigated from a variety of different angles, and these are by no means restricted to scientific analyses of an individual slag fragment. It is of pivotal importance to keep the archaeological context information in mind. In my dissertation the archaeology held to the key to the understanding of the whole smelting enterprise conducted at the site!
The scientific analysis starts out with the documentation and description of the macroscopic appearance. The next step is a visual examination by means of a stereo-microscope, which should assist in thesample preparation for light and electron microscopical investigations. By means of polarising reflected light microscopy, also known as ore microscopy, a wealth of information can be collected:
- general state of preservation
- texture
- number of phases that are present
- identification of phase
After this stage samples can be scrutinised by means of electron microscopy, such as scanning electron microscopes, electron micro-probe, or other micro-analytical tools which provide us with information that lead to the identification of slag phases.
A parallel approach is the determination of the bulk chemistry of the samples, as this information is needed to understand the chemical interaction between the individual furnace charge components. These analyses can be made by x-ray fluorescence or mass spectroscopy. It is good practice to investigate samples by optical means first: Areas affected by corrosion and subsequently suffered from chemical alteration can thus be excluded from analysis.
How to distinguish slags?
Slags may ultimately only distinguished by their chemistry and phase composition. A macroscopic differentiation may only be correct on regional scales and where it is certain that similar processes had been in effect.
Read more about slag check this. be sure to leave a comment inf you like what you are reading. Comments and criticism are appreciated.







October 18th, 2018 at 14:25
An iron furnace has been discovered on an archaeological site in the Northern Shenandoah Valley of Virginia.
A section of furnace wall was Tl dated at the University of Washington.
Testing shows the iron furnace was used 150 AD.
I cannot find an archeometallurgy expert that will investigate.
I am happy to forward test results.
Please contact me.
Thank you,
Adam Arkfeld
March 21st, 2019 at 17:13
I prefer to work with you
March 21st, 2019 at 20:31
I can now clearly understand the whole process of slag analysis